Embedded Machine Learning
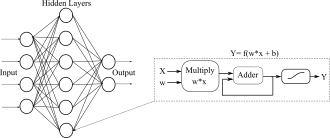
Machine learning algorithms, specifically neural-networks, have been used extensively to classify, predict and capture complex relationships. In the case of biomedical devices, these algorithms are used to make the devices robust to variability and increase their accuracy. However, currently these algorithms require power-hungry computational resources to operate and are not suitable for implantable or wearable devices. Thus, it is important to explore and develop novel machine learning algorithms and hardware that can efficiently infer data in real-time.
One of the major sources of power consumption in an embedded machine learning platform is communication between memory and compute. Hence, I am investigating embedded machine learning architectures, and circuits and system techniques that reduce the energy associated with fetching of weights from the memory, algorithms that enable reuse of weights and biases, and systems that do not have explicit demarcation between memory and compute. For instance, in my previous work I have shown that a crossbar array composed of FG transistors can be used for vector matrix multiplication. The charge stored on the floating node, programmed using hot-electron injection process, is used as the weight of the vector-matrix multiplication. Such crossbar arrays are highly energy-efficient and dense since they have no explicit distinction between memory and compute during inference (paper).
In the case of an all-digital neural network architecture, the communication can be made efficient by representing the weights and biases with reduced precision without affecting the overall accuracy of the network. The power consumption associated with storing and fetching the weights is more than halved when the weights are represented with a 16-bit accuracy as opposed to 32-bit accuracy without affecting the precision of the network.
One of the major sources of power consumption in an embedded machine learning platform is communication between memory and compute. Hence, I am investigating embedded machine learning architectures, and circuits and system techniques that reduce the energy associated with fetching of weights from the memory, algorithms that enable reuse of weights and biases, and systems that do not have explicit demarcation between memory and compute. For instance, in my previous work I have shown that a crossbar array composed of FG transistors can be used for vector matrix multiplication. The charge stored on the floating node, programmed using hot-electron injection process, is used as the weight of the vector-matrix multiplication. Such crossbar arrays are highly energy-efficient and dense since they have no explicit distinction between memory and compute during inference (paper).
In the case of an all-digital neural network architecture, the communication can be made efficient by representing the weights and biases with reduced precision without affecting the overall accuracy of the network. The power consumption associated with storing and fetching the weights is more than halved when the weights are represented with a 16-bit accuracy as opposed to 32-bit accuracy without affecting the precision of the network.
Brain-Machine Interface
In the United States there are about 17,700 new cases per year of Spinal Cord Injury (SCI). SCI results in a partial or total loss of motor function. Brain-Machine Interfaces (BMI) have the potential to increase independence and improve quality of life in SCI patients by reading out neural signals and mapping them onto control signals for assistive devices.
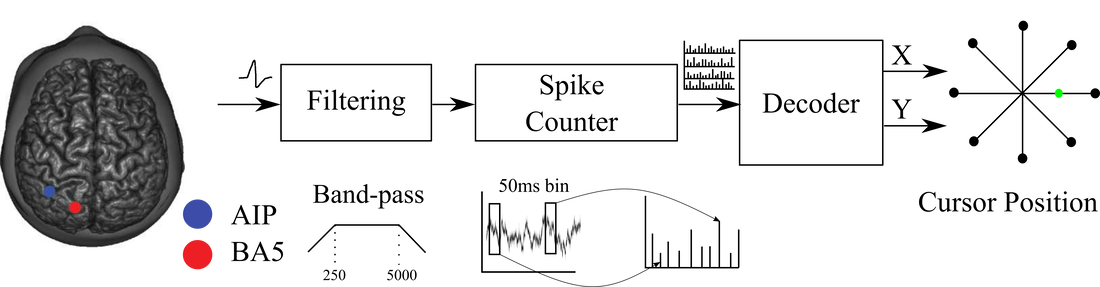
Data used in our is recorded from the posterior parietal cortex of a tetraplegic human research participant. Cells in this region have been shown to encode the goal of movements and are also involved in sensorimotor integration and high-level motor planning. These findings suggest that neural signals recorded from the parietal lobe could be useful for a variety of BMI tasks.
In out initial work we compare the machine learning algorithms such as Recurrent Neural Networks with conventional decoding approaches such as kalman filter (paper). In addition we also believe that an integral part of developing robust BMI decoders will be to use features of the neural data that convey information stably over time. Hence, we are also investigating various feature extraction techniques to increase the accuracy of the decoder over multiple days. This work was accepted in NeurIPS 2019 (paper).
Currently state-of-art-decoders are implemented using a desktop computer consuming several watts of power (a typical desktop consumes 60 to 300 watts of power). Such a system is not optimized for real-time processing outside of a clinical setting. Part of making BMI systems clinically relevant is to design and develop area- and power-efficient hardware for decoding kinematics such that these systems can be implanted or worn on the body. Towards this goal I am also investigating novel architectures to compute these machine learning algorithms on an embedded or wearable platforms.
Data used in our is recorded from the posterior parietal cortex of a tetraplegic human research participant. Cells in this region have been shown to encode the goal of movements and are also involved in sensorimotor integration and high-level motor planning. These findings suggest that neural signals recorded from the parietal lobe could be useful for a variety of BMI tasks.
In out initial work we compare the machine learning algorithms such as Recurrent Neural Networks with conventional decoding approaches such as kalman filter (paper). In addition we also believe that an integral part of developing robust BMI decoders will be to use features of the neural data that convey information stably over time. Hence, we are also investigating various feature extraction techniques to increase the accuracy of the decoder over multiple days. This work was accepted in NeurIPS 2019 (paper).
Currently state-of-art-decoders are implemented using a desktop computer consuming several watts of power (a typical desktop consumes 60 to 300 watts of power). Such a system is not optimized for real-time processing outside of a clinical setting. Part of making BMI systems clinically relevant is to design and develop area- and power-efficient hardware for decoding kinematics such that these systems can be implanted or worn on the body. Towards this goal I am also investigating novel architectures to compute these machine learning algorithms on an embedded or wearable platforms.
Real-time monitoring of vital/physiological signals
State-of-the-art wearable devices rely heavily on cloud computers or remote servers to analyze the data they generate. Energy and bandwidth required for such wireless communication is large and is set to grow with the increase in number of wearable devices. This is particularly true for devices that are used for sensing, monitoring, and analyzing biological signals since the amount of data generated is large. Hence, there is a need for investigating and developing circuits and system that can efficiently process and analyze biological signals in real-time.
Heart disease (HD) is the primary reason for death in the U.S. and health care expenditure for HD imposes the largest burden on the total national health spending. Furthermore, it is expected that by 2035, the percentage of the U.S. population having at least one HD will rise to 45%. Early diagnosis, and timely management of HD can potentially lower risk of complications, thus improving quality-of-life. However, current HD diagnosis approach is reactive and therefore inappropriate for early diagnosis: Only after symptoms occur patients visit the clinic, where expensive procedures are used for diagnosis. In contrast, a proactive approach, where people are ubiquitously monitored at home for timely detection of signs of abnormalities before serious symptoms manifest, can increase the rate of early diagnosis. Such proactive approach can also facilitate treatment being tuned to changes in patients’ physiology, thereby potentially increasing treatment success rates and reducing the frequency of visits to the clinic as well as the healthcare costs.
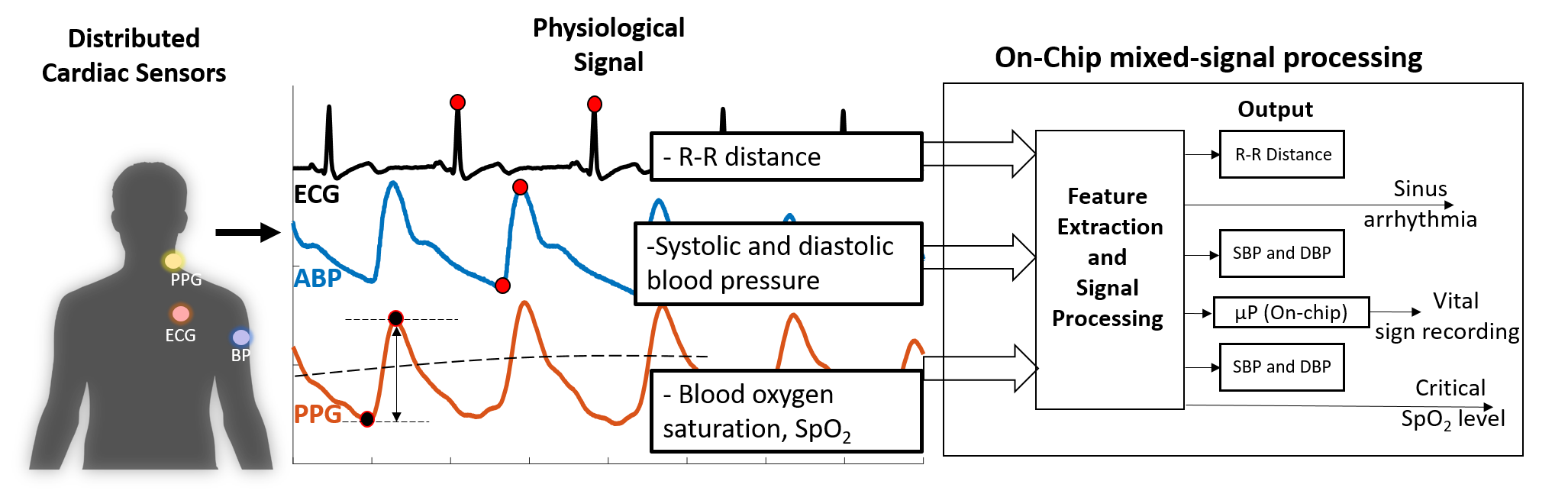
In a recent study, we demonstrate that a re-programmable mixed-signal systems can be used to monitor several hemodynamic features such as heart rate, blood pressure, and blood oxygen saturation derived from electrocardiography, arterial blood pressure, and photoplethysmography signals in real-time with substantially low-power consumption (paper).
In a recent study, we demonstrate that a re-programmable mixed-signal systems can be used to monitor several hemodynamic features such as heart rate, blood pressure, and blood oxygen saturation derived from electrocardiography, arterial blood pressure, and photoplethysmography signals in real-time with substantially low-power consumption (paper).